Steric and electronic effects in stabilizing allyl-palladium complexes of “P–N–P” ligands, X2PN(Me)PX2 (X = OC6H5 or OC6H3Me2-2,6) - ScienceDirect

PDF) Formation of Palladium- and Platinum-Substituted Fulvenes by Cyclopentadienyl Activation in a Formal Insertion Reaction | Tomás Belderraín and Pedro Valerga - Academia.edu

Fei-Phos ligand-controlled asymmetric palladium-catalyzed allylic substitutions with structurally diverse nucleophiles: scope and limitations - RSC Advances (RSC Publishing) DOI:10.1039/C6RA09657C

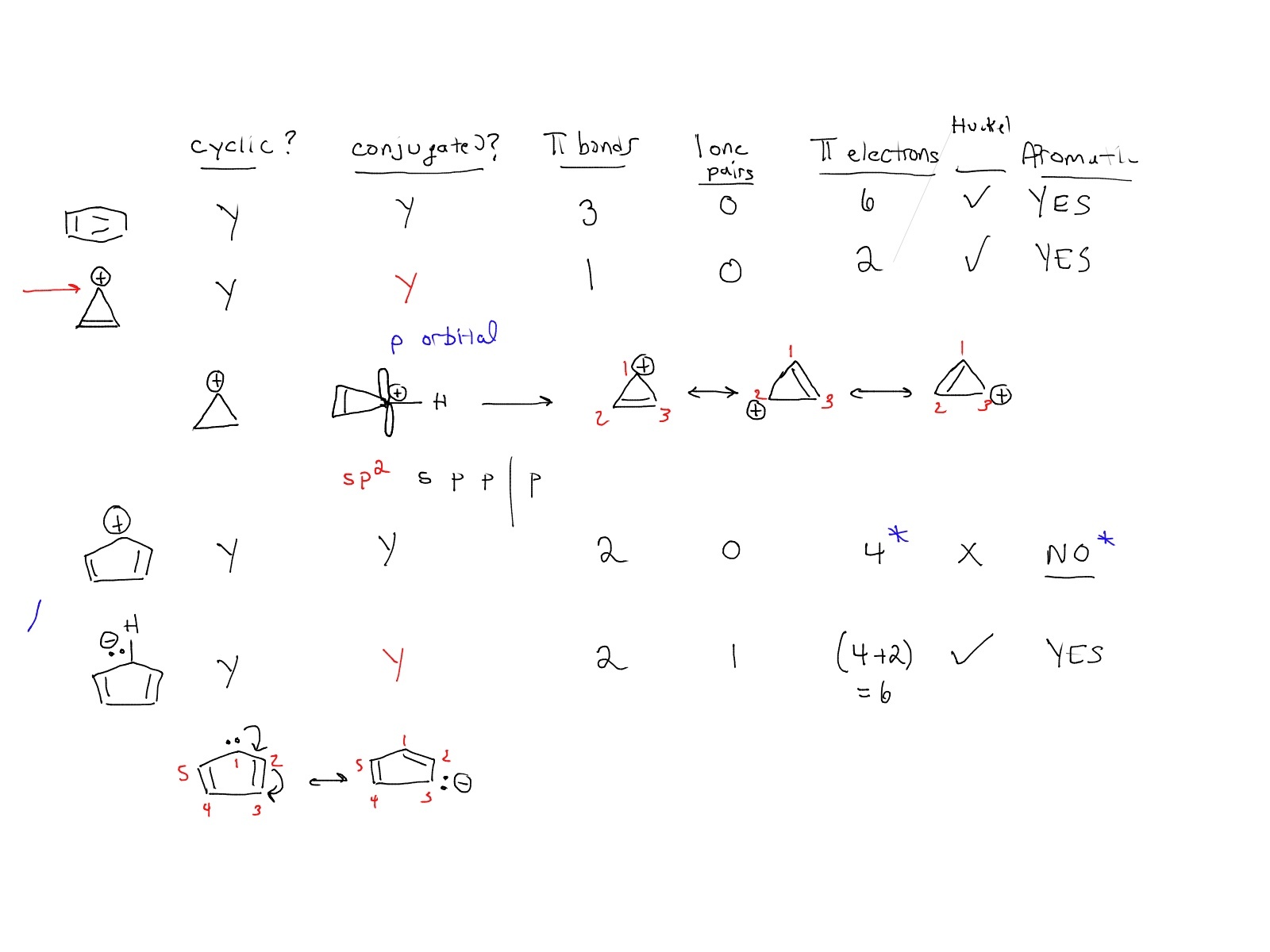
Account for the formation of the following product, including the cis stereochemistry at the ring junction. | Homework.Study.com
Combined high-pressure experimental and kinetic modeling study of cyclopentene pyrolysis and its reactions with acetylene

Telomerization of 1,3‐Butadiene with Carbon Dioxide: A Highly Efficient Process for δ‐Lactone Generation - Balbino - 2018 - ChemCatChem - Wiley Online Library
![PDF] The chemistry of ruthenium, rhodium, palladium, osmium, iridium and platinum | Semantic Scholar PDF] The chemistry of ruthenium, rhodium, palladium, osmium, iridium and platinum | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f5f9b71065581b62f056671b6377a83ddfd985b6/54-Table11-1.png)
PDF] The chemistry of ruthenium, rhodium, palladium, osmium, iridium and platinum | Semantic Scholar

Palladium Complexes of Methylene-Bridged P-Stereogenic, Unsymmetrical Diphosphines | Organometallics
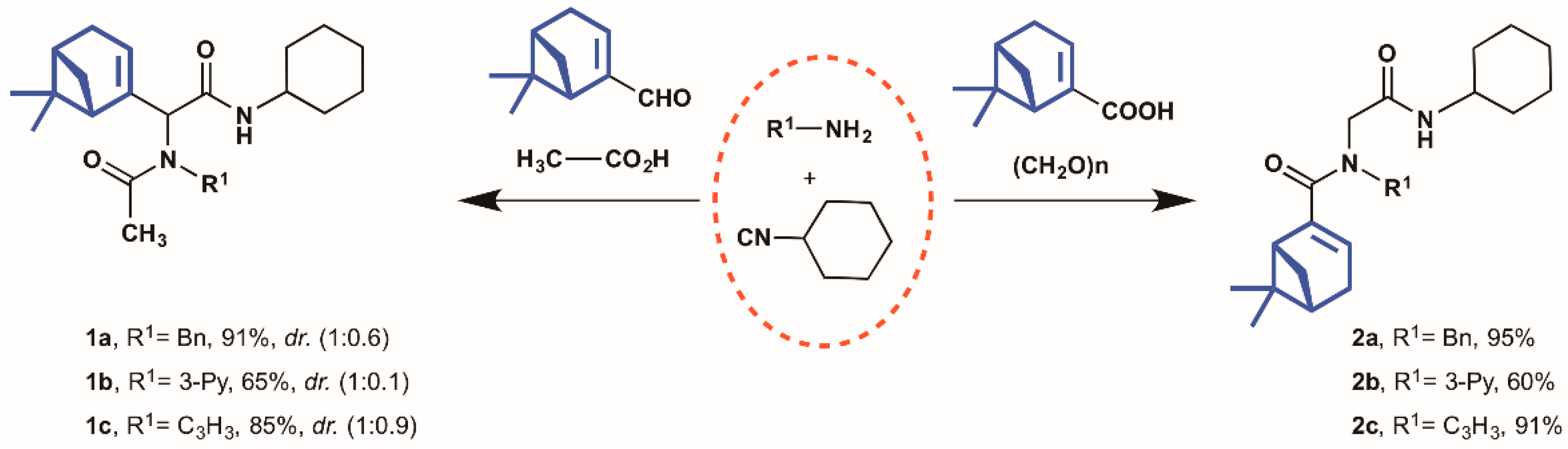
Molecules | Free Full-Text | Synthesis and Cytotoxic Analysis of Novel Myrtenyl Grafted Pseudo-Peptides Revealed Potential Candidates for Anticancer Therapy
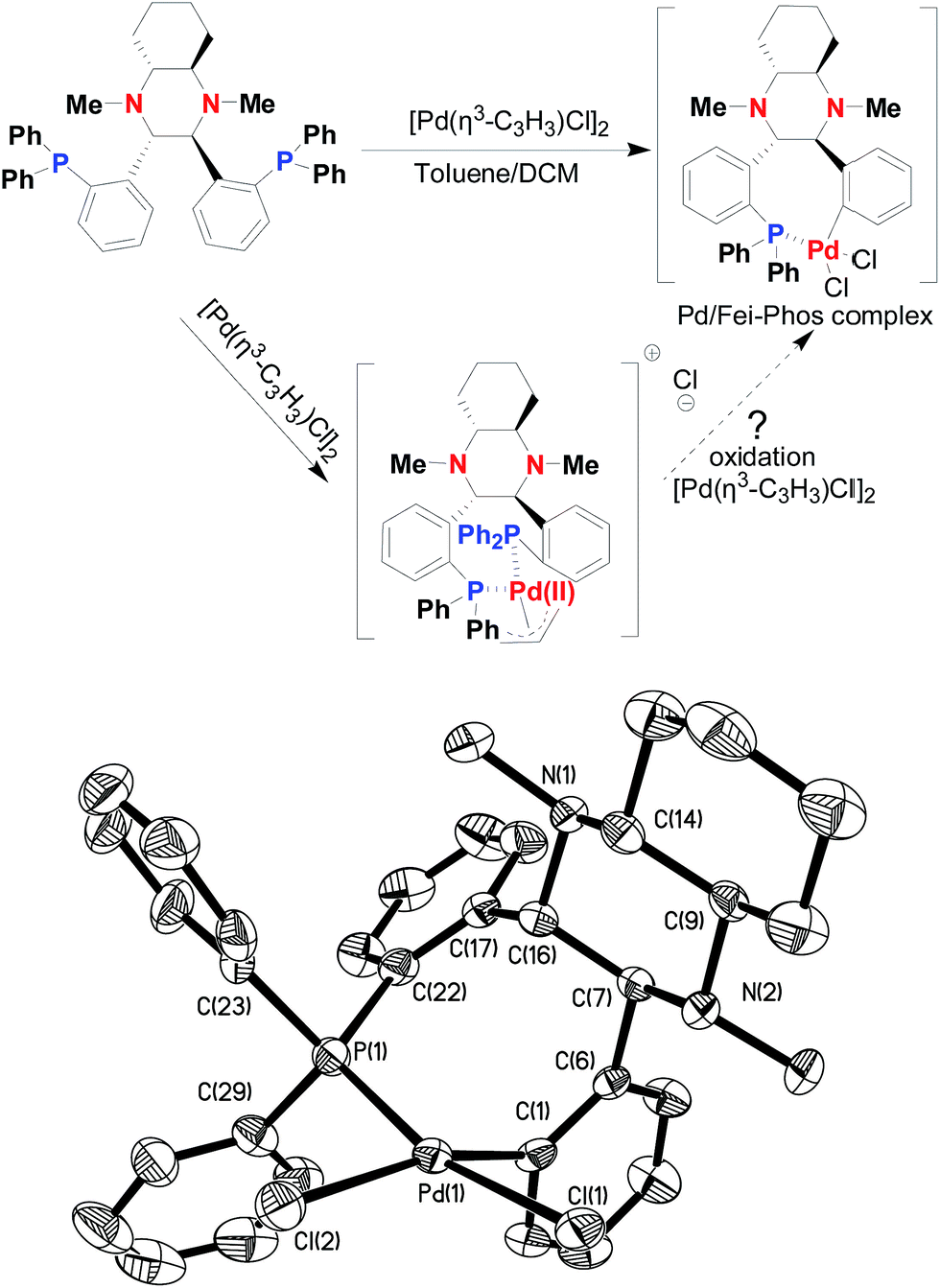
A mechanistic study on multifunctional Fei-Phos ligand-controlled asymmetric palladium-catalyzed allylic substitutions - RSC Advances (RSC Publishing) DOI:10.1039/C6RA15665G

PDF) Allyl Amination of Phosphinoquinoline Allyl Complexes of Palladium. Influence of the Allyl Hapticity on the Reaction Rate and Regiochemistry | Valerio Bertolasi - Academia.edu
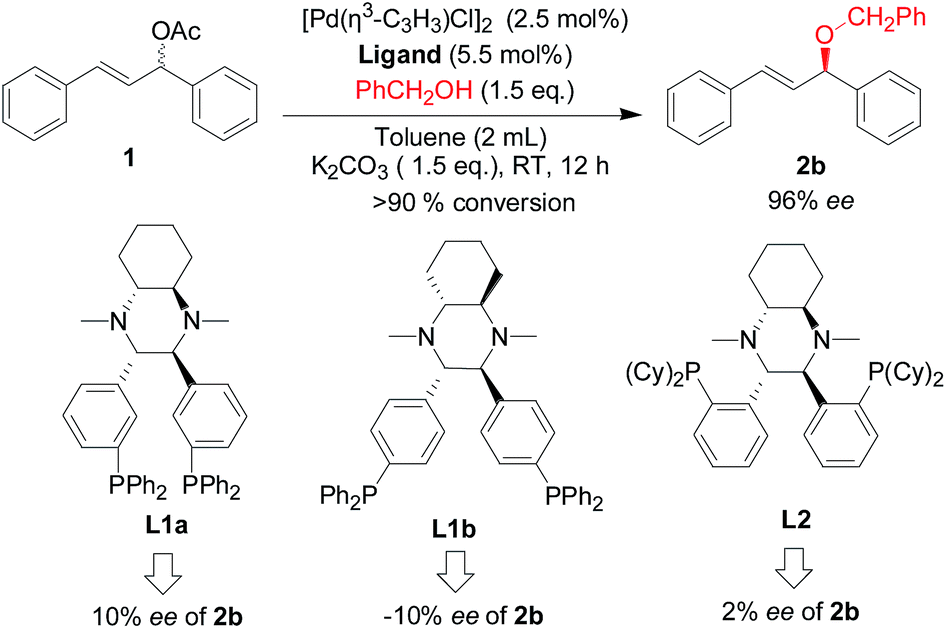
Fei-Phos ligand-controlled asymmetric palladium-catalyzed allylic substitutions with structurally diverse nucleophiles: scope and limitations - RSC Advances (RSC Publishing) DOI:10.1039/C6RA09657C

A Ketimide-Stabilized Palladium Nanocluster with a Hexagonal Aromatic Pd7 Core | Inorganic Chemistry
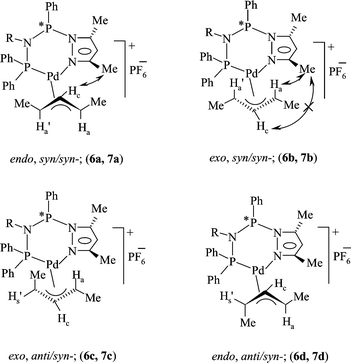
Palladium( ii ) allyl complexes of chiral diphosphazane ligands: ambident coordination behaviour and stereodynamic studies in solution - Dalton Transactions (RSC Publishing) DOI:10.1039/B207447H

PDF) Stereochemical study of natural sesquiterpenic complexes by X-rays | Larbi El Firdoussi - Academia.edu

A Detailed Study of Acetate-Assisted C–H Activation at Palladium(IV) Centers | Journal of the American Chemical Society
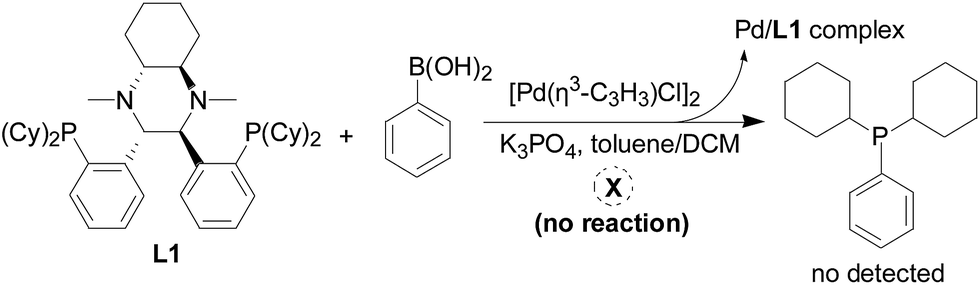
A mechanistic study on multifunctional Fei-Phos ligand-controlled asymmetric palladium-catalyzed allylic substitutions - RSC Advances (RSC Publishing) DOI:10.1039/C6RA15665G

A Detailed Study of Acetate-Assisted C–H Activation at Palladium(IV) Centers | Journal of the American Chemical Society

Steric and electronic effects in stabilizing allyl-palladium complexes of “P–N–P” ligands, X2PN(Me)PX2 (X = OC6H5 or OC6H3Me2-2,6) - ScienceDirect

A Detailed Study of Acetate-Assisted C–H Activation at Palladium(IV) Centers | Journal of the American Chemical Society
![PDF) Convenient Synthesis of Alkenyl-, Alkynyl-, and Allenyl-Substituted Imidazo[1,2-a]pyridinesvia Palladium-Catalyzed Cross-Coupling Reactions PDF) Convenient Synthesis of Alkenyl-, Alkynyl-, and Allenyl-Substituted Imidazo[1,2-a]pyridinesvia Palladium-Catalyzed Cross-Coupling Reactions](https://i1.rgstatic.net/publication/230129898_Convenient_Synthesis_of_Alkenyl-_Alkynyl-_and_Allenyl-Substituted_Imidazo12-apyridinesvia_Palladium-Catalyzed_Cross-Coupling_Reactions/links/5b7edd9a92851c1e122ce63f/largepreview.png)
PDF) Convenient Synthesis of Alkenyl-, Alkynyl-, and Allenyl-Substituted Imidazo[1,2-a]pyridinesvia Palladium-Catalyzed Cross-Coupling Reactions